Leukocyte Dynamics (Kastenmüller Lab)
Our lab focuses on cell-cell interactions, cellular localization and migration between and within tissues. To address such questions we combine classical immunological assays like multicolour flow cytometry with cutting edge microscopy including 2-photon imaging of live animals, confocal analysis of tissue section and whole mount preparations to study cellular immune responses in the context of infections. Central questions currently addressed in our lab include:
- Function and development cytotoxic CD8+ T cells
- Migration and dynamics of Dendritic Cells within tissues
- Immune defense against viral and bacterial infections
- Interface between innate and adaptive immune system
- Intercellular communication
- Development of new animal model
We are a young dynamic team and with a high technical expertise and a broad research interest. We are involved in various collaborations with national and international research teams.
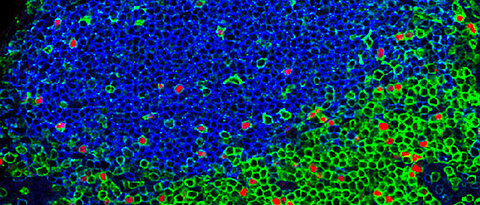
The lymphatic network that transports interstitial fluid and antigens to lymph nodes forms a conduit system that can be hijacked by invading pathogens to achieve systemic spread unless dissemination is blocked in the lymph node itself. We found that a network of diverse lymphoid cells (natural killer cells, gd T cells, natural killer T cells, and innate-like CD8+ T cells) are spatially prepositioned close to lymphatic sinus-lining macrophages where they can rapidly and efficiently receive inflammasome-generated IL-18 and additional cytokine signals from the pathogen-sensing phagocytes. This leads to rapid IFNg secretion by the strategically positioned innate lymphocytes, fostering antimicrobial resistance in the macrophage population. Interference with this innate immune response loop allows systemic spread of lymphborne bacteria. Our findings extends our understanding of the functional significance of cellular positioning and local intercellular communication within lymph nodes while emphasizing the role of these organs as highly active locations of innate host defense.
(Kastenmüller W et al. Cell. 2012 Sep 14;150(6):1235-48. Gasteiger G et al. Immunol Rev. 2016 May;271(1):200-20)
After an infection, the immune system generates long-lived memory lymphocytes whose increased frequency and altered state of differentiation enhance host defense against reinfection. The spatial distribution of memory cells was found to contribute to their protective function. Tissue-resident memory CD8+ T cells reside in peripheral tissue at sites of initial pathogen encounter. We have shown that within lymph nodes (LNs), memory CD8+ T cells are also concentrated near peripheral entry portals of lymph-borne pathogens, promoting rapid engagement of infected sinus-lining macrophages. A feed-forward CXCL9- dependent circuit provides additional chemotactic cues that further increase local memory T cell density. Memory CD8+ T cells also produce effector responses to local cytokine triggers, but their dynamic behavior differe from that seen after antigen recognition. We have revealed the distinct localization and dynamic behavior of naive versus memory T cells within LNs and how these differences contribute to host defense.
(Kastenmuller W et al. Immunity. 2013 Mär 21;38(3):502-13, Qi et al. Annu Rev Cell Dev Biol. 2014;30:141-67.)
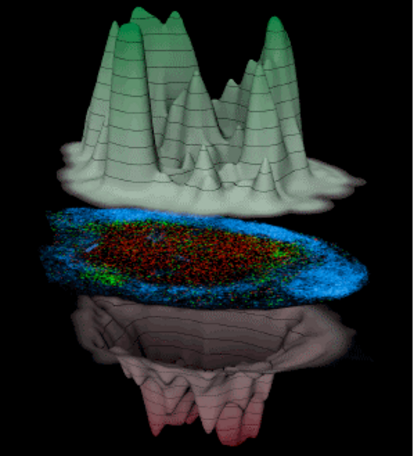
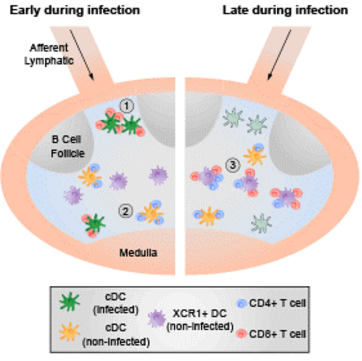
Host defense against viruses and intracellular parasites depends on effector CD8+ T cells whose optimal clonal expansion, differentiation, and memory properties require helper signals from CD4+ T cells. We have addressed the role of dendritic cell (DC) subsets in initial activation of the two T cell types (CD4 and CD8 T cells) and their co-operation. Surprisingly, initial priming of CD4+ and CD8+ T cells was spatially segregated within the lymph node and occurred on different DC with temporally distinct patterns of antigen-presentation via MHCI vs. MHCII molecules. DC that co-present antigen via both MHC molecules were detected at a later stage (>24h post infection); these XCR1+-DC are the critical platform involved in CD4+ T cell augmentation of CD8+ T cell responses. With these findings we delineated the complex choreography of cellular interactions underlying effective cell-mediated anti-viral responses, with implications for basic DC subset biology as well as for translational application to the development of vaccines that evoke optimal T cell immunity.
(Eickhoff S et al. Cell 2015 Sep 10;162(6):1322-37 und Borst J. et al. Nat Rev. Immunol. 2018 Jul 29.)
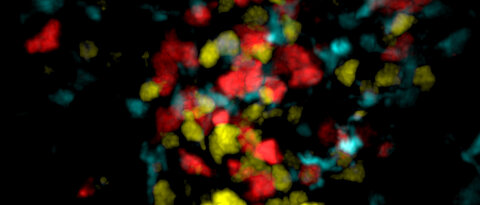
Tumors and chronic infections result in sustained antigen exposure, which promotes impaired functional responsiveness in T cells referred to as exhaustion. Checkpoint immunotherapy can induce the reinvigoration of cellular immunity by activating a recently identified precursor of exhausted T (TPEX) cell population. Conventional dendritic cells (cDC) likely play an essential role for directing TPEX cell differentiation and for regulating their maintenance. In this project, we aimed to elucidate, where and when cDC - TPex cell interactions take place and which cDC subsets are involved. To address these questions, we mapped the transcriptional differentiation trajectory of TPEX cell subsets towards terminally exhausted T (TEX) cells, identified transcriptionally distinct subpopulations and defined their localization in the spleen during chronic viral infection. We found that cDC1, a specialized subset of dendritic cells, colocalized with TPEX cells and regulated their maintenance by promoting the functionality of stromal cells within a specialized TPEX cell nice. Additionally, during αPD-L1 treatment, the splenic cDC1 network was significantly reorganized at the marginal zone, a site of TPEX cell activation and differentiation. As a consequence, viral control during check-point immunotherapy and cellular integrity of the marginal zone depended on the presence of cDC1. Our data reveal the location of cDC1 - TPEX cell interactions and provide direct evidence that cDC1 are essential to maintain TPEX cells whilst guiding their differentiation to control viral replication. These findings reveal mechanisms of TPEX cell differentiation and maintenance and therefore have critical implications for the improvement of checkpoint immunotherapy. Currently, we are focusing on the function of other antigen-presenting cells like cDC2 and monocytes and elucidate their role in promoting immunopathology.
Lymph nodes (LNs) function as immunological filters for lymph borne pathogens and are essential organs for initiating adaptive immunity after infection and vaccination. Lymphatic transport of molecules and migration of myeloid cells to LNs are fundamental in this process as they continuously inform lymphocytes on changes in the drained tissues. In this project we asked whether other cell types besides dendritic cells (cDC) migrate via the lymph and thereby establish another cellular link between tissues and lymph node function and immune activation. We focused on unconventional T cells (UTC) that consist of three major lineages – gamma delta (γδ), MR1-restricted (e.g., MAIT), and CD1d-restricted T cells (e.g., NKT) and all display a TCR repertoire of limited diversity. These cells are selected by non-polymorphic antigen-presenting molecules like CD1 and MR1, and typically recognize endogenous lipids or microbial metabolites bound to these surface proteins.
They differentiate into polarized effector cells already in the thymus with properties similar to specialized CD4 T cell subsets and seed organs in developmental waves. Here, we show that these tissue-derived UTC migrate via the lymphatic route to locally draining LNs similar to cDC. Because each tissue harbors a distinct spectrum of UTC with locally adapted differentation states and distinct TCR repertoires, every draining LN is thus populated by a distinctive tissue-determined mix of these lymphocytes. Lymphatic migration of UTC is therefore a key determinant of tissue-specific immunity, which is characteristically initiated in different LNs. This results in differences in immune responses depending on the site of infection or vaccination route, which in turn has significant implications for vaccination strategies and immunotherapeutic approaches.
Kastenmueller link pubmed: https://pubmed.ncbi.nlm.nih.gov
Selected Articles
- A distinct priming phase regulates CD8 T cell immunity by orchestrating paracrine IL-2 signals. Katarzyna Jobin, Deeksha Seetharama, Lennart Rüttger, Cloe Fenton, Ekaterina Kharybina, Annerose Wirsching, Anfei Huang, Konrad Knöpper, Tsuneyasu Kaisho, Dirk H. Busch, Martin Vaeth, Antoine Emmanuel Saliba, Frederik Graw, Alain Pulfer, Santiago F. Gonzalez, Dietmar Zehn, Yinming Liang, Milas Ugur, Georg Gasteiger and Wolfgang Kastenmüller. Science 388,eadq1405(2025).DOI:10.1126/science.adq1405
- Resident regulatory T cells reflect the immune history of individual lymph nodes. Kaminski A, Hager FT, Kopplin L, Ticconi F, Leufgen A, Vendelova E, Rüttger L, Gasteiger G, Cerovic V, Kastenmüller W, Pabst O, Ugur M. Published in Science Immunology 2023. DOI: 10.1126/sciimmunol.adj5789
- Lymph node medulla regulates the spatiotemporal unfolding of resident dendritic cell networks. Ugur M, Labios RJ, Fenton C, Knöpper K, Jobin K, Imdahl F, Golda G, Hoh K, Grafen A, Kaisho T, Saliba AE, Grün D, Gasteiger G, Bajénoff M, Kastenmüller W., Immunity. 2023 Aug 8;56(8):1778-1793.e10. doi: 10.1016/j.immuni.2023.06.020. Epub 2023 Jul 17.
- CD4+ T cell-induced inflammatory cell death controls immune-evasive tumours, Bastian Kruse, Anthony C. Buzzai, Naveen Shridhar, Andreas D. Braun, Susan Gellert, Kristin Knauth, Joanna Pozniak, Johannes Peters, Paulina Dittmann, Miriam Mengoni, Tetje Cornelia van der Sluis, Simon Höhn, Asier Antoranz, Anna Krone, Yan Fu, Di Yu, Magnus Essand, Robert Geffers, Dimitrios Mougiakakos, Sascha Kahlfuß, Hamid Kashkar, Evelyn Gaffal, Francesca M. Bosisio, Oliver Bechter, Florian Rambow, Jean-Christophe Marine, Wolfgang Kastenmüller, Andreas J. Müller & Thomas Tüting, nature. 2023; June 14; doi: 10.1038/s41586-023-06199-x
- Lymphatic migration of unconventional T cells promotes site-specific immunity in distinct lymph nodes. Ataide MA, Knöpper K, Cruz de Casas P, Ugur M, Eickhoff S, Zou M, Shaikh H, Trivedi A, Grafen A, Yang T, Prinz I, Ohlsen K, Gomez de Agüero M, Beilhack A, Huehn J, Gaya M, Saliba AE, Gasteiger G, Kastenmüller W, Immunity. 2022 Aug 17:S1074-7613(22)00354-5. ; doi: 10.1016/j.immuni.2022.07.019
- Type 1 conventional dendritic cells maintain and guide the differentiation of precursors of exhausted T cells in distinct cellular niches. Dähling S, Mansilla AM, Knöpper K, Grafen A, Utzschneider DT, Ugur M, Whitney PG, Bachem A, Arampatzi P, Imdahl F, Kaisho T, Zehn D, Klauschen F, Garbi N, Kallies A, Saliba AE, Gasteiger G, Bedoui S, Kastenmüller W. Immunity. 2022 Apr 12; 55(4):656-670.e8 ; doi: 10.1016/j.immuni.2022.03.006
- BATF3 programs CD8+ T cell memory. Ataide MA, Komander K, Knöpper K, Peters AE, Wu H, Eickhoff S, Gogishvili T, Weber J, Grafen A, Kallies A, Garbi N, Einsele H, Hudecek M, Gasteiger G, Hölzel M, Vaeth M, Kastenmüller W. Nat Immunol. 2020 Nov;21(11):1397-1407. doi: 10.1038/s41590-020-0786-2.Open Access
- Lymphatic Endothelial Cells Are Essential Components of the Subcapsular Sinus Macrophage Niche. Mondor I, Baratin M, Lagueyrie M, Saro L, Henri S, Gentek R, Suerinck D, Kastenmuller W, Jiang JX, Bajénoff M. Immunity. 2019 Jun 18;50(6):1453-1466.e4. doi: 10.1016/j.immuni.2019.04.002
- Perforin inhibition protects from lethal endothelial damage during fulminant viral hepatitis. Welz M, Eickhoff S, Abdullah Z, Trebicka J, Gartlan KH, Spicer JA, Demetris AJ, H. Akhlaghi H, Anton M, Manske K, Zehn D, Nieswandt B, Kurts C, Trapani JA, Knolle P, Wohlleber D, Kastenmüller W. Nat Comm 2018 Nov 15;9(1):4805. doi: 10.1038/s41467-018-07213-x.
- CD8+ T Cells Orchestrate pDC-XCR1+ Dendritic Cell Spatial and Functional Cooperativity to Optimize Priming. Brewitz A, Eickhoff S, Dähling S, Quast T, Bedoui S, Kroczek RA, Kurts C, Garbi N, Barchet W, Iannacone M, Klauschen F, Kolanus W, Kaisho T, Colonna M, Germain RN, Kastenmüller W. Immunity 2017 Feb 21;46(2):205-219. doi: 10.1016/j.immuni.2017.01.003.
- Robust Anti-viral Immunity Requires Multiple Distinct T Cell-Dendritic Cell Interactions. Eickhoff S, Brewitz A, Gerner MY, Klauschen F, Komander K, Hemmi H, Garbi N, Kaisho T, Germain RN, Kastenmüller W. Cell 2015 Sep 10;162(6):1322-37. doi: 10.1016/j.cell.2015.08.004.
Selected Review Articles
- Concepts of GPCR-controlled navigation in the immune system. Lämmermann T, Kastenmüller W. Immunol Rev. 2019 May;289(1):205-231. doi: 10.1111/imr.12752
- CD4+ T cell help in cancer immunology and immunotherapy. Borst J, Ahrends T, Bąbała N, Melief CJM, Kastenmüller W. Nat Rev Immunol. 2018 Oct;18(10):635-647. doi: 10.1038/s41577-018-0044-0.
- Lymph node - an organ for T-cell activation and pathogen defense. Gasteiger G, Ataide M, Kastenmüller W. Immunol Rev. 2016 May;271(1):200-20. doi: 10.1111/imr.12399.
- Dendritic cell-targeted vaccines - hope or hype? Kastenmuller W, Kastenmuller K, Kurts C, Seder RA. 2014. Nat Rev Immunol 2014 Oct;14(10):705-11. doi: 10.1038/nri3727.
- Spatiotemporal Basis of Innate and Adaptive Immunity in Secondary Lymphoid Tissue. Qi H, Kastenmuller W, Germain RN. 2014. Annu Rev Cell Dev Biol 2014;30:141-67. doi: 10.1146/annurev-cellbio-100913-013254.
- Foxp3+ Regulatory T-cells and IL-2: The Moirai of T-cell Fates? Gasteiger G, Kastenmuller W.. Front Immunol 2012 3: 179. doi: 10.3389/fimmu.2012.00179.
- DFG Funding for Immunology Research in Würzburg - 12.12.2023
- Immune guardians in the lymph node - 26.10.2023
- Immune Cells in Single File - 17.07.2023
- New Players in the Immune Response - 23.08.2022
- Why Sentinel Cells are so Important - 01.04.2022
- Start-stop System of Hunting Immune Cells - 18.06.2021
- Memory training for the immune system - 28.09.2020

Defne Akkar


Samuel dos Santos Oliveira


Kathrin Hoh

Dr. Anfei Huang







Dr. Milas Ugur


Dr. Xiaoli Yang