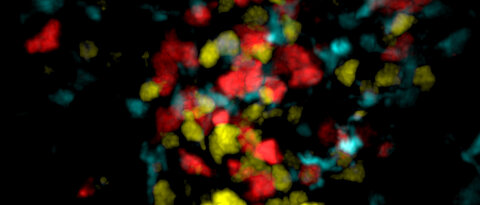
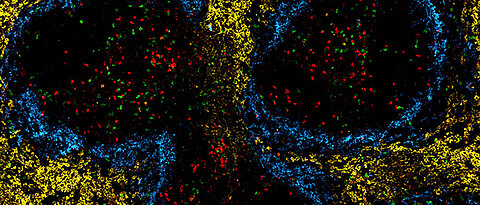
Metabolism and Immune Cell Signalling (Vaeth Lab)
Immunological tolerance is the balance between immunity to infection and autoimmunity, dysregulation of which can result in severe immunological pathologies. Our laboratory investigates the molecular regulation of (patho-) physiological immune responses with a particular focus on the metabolic programming of lymphocytes. Over the last decade, it became obvious that ‘immunometabolism’ not only provides chemical energy and building blocks for the clonal expansion and effector function of lymphocytes, but also governs the epigenetic programming of immune cells. Thus, a better understanding of the metabolic-epigenetic regulatory circuits in immune cells promises new therapeutic avenues to treat immune-related pathologies, such as infection, cancer or autoimmunity.
Unlike most other tissues, immune cells are not sessile but patrol and populate all organs in the body. To cope with the varying metabolic conditions in different (non-lymphoid) tissues, immune cells are equipped with an extensive enzymatic repertoire. This versatility of leukocytes and the notion that cellular metabolism is intrinsically linked to the outcome of complex immune responses established ‘immunometabolism’ as an independent and interdisciplinary research field. A plethora of catabolic and anaplerotic reactions are directly linked to signal transduction, epigenetic modifications and the control of gene expression. Thus, the interrelation of cellular metabolism, signaling and epigenetic imprinting constitutes an intra-cellular network that controls the fate and function of immune cells.
We study various aspects of this network in the immune system across scales by combining functional studies in transgenic animal models with (single-cell) transcriptomic, metabolomic and epigenetic analyses. A better understanding how metabolism controls the complex functions of the immune system will not only provide novel therapeutic concepts to treat inflammatory and autoimmune diseases but will also help to exploit the inherent capacity of leukocytes to combat pathogens and cancer.
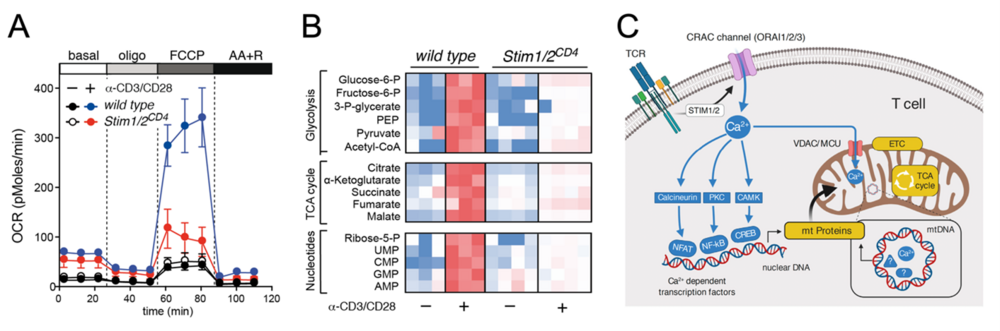
We showed previously that cell cycle entry and clonal expansion of virus-specific T cells is controlled by calcium signaling and transcription factors of the nuclear factor of activated T cells (NFAT) family. NFAT controls the expression of nutrient transporters and metabolic enzymes to support cellular growth and proliferation of T cells. To meet their bioenergetic demand, effector T cells rewire their metabolic machinery to aerobic glycolysis to generate ATP and building blocks for the biosynthesis of macromolecules. By contrast, memory and regulatory T cells are less glycolytic and rely, instead, on oxidative phosphorylation (OXPHOS) and fatty acid oxidation (FAO). The goal of this project is to define how calcium and NFAT signaling contributes to the specific metabolic phenotypes of different immune cell subsets. Specifially, we pursue the following goals:
- To delineate the shared and redundant functions of store-operated calcium entry (SOCE), calcineurin signaling and NFAT-mediated gene expression
- To define the role of different NFAT family members and their isoforms during the metabolic reprogramming of lymphocytes
- To investigate the reciprocal regulation of SOCE and NFAT signaling by intermediary metabolites
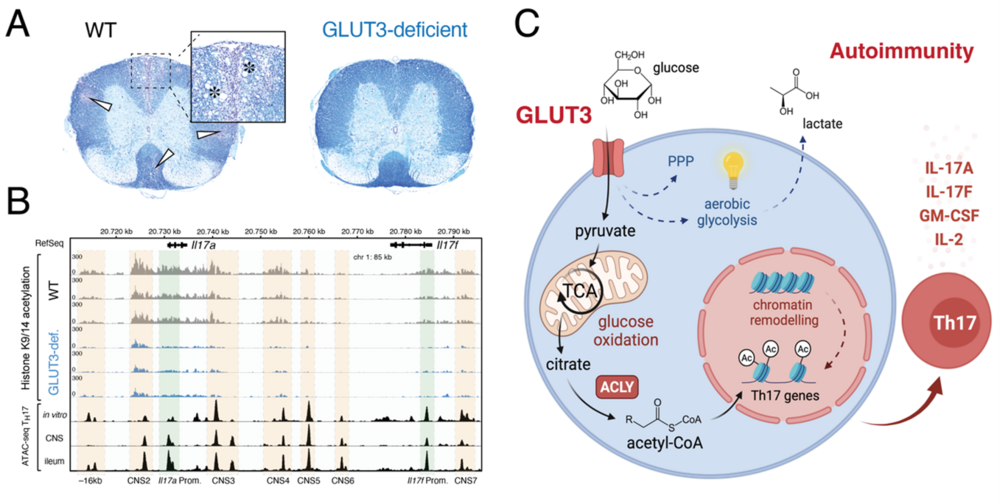
Glucose is the universal fuel for metabolic processes in all tissues of the body. Immune cells are no exception and utilize two main transporters to utilize glucose: GLUT1 and GLUT3. In this project we focus on the glucose transporter GLUT3, because of its high expression in effector, tissue-resident and regulatory T cells. Unlike the universal hexose transporter GLUT1, GLUT3 seems dispensable for energy production and clonal expansion of T cells, but is crucial for their pathogenicity in models of autoimmunity. Metabolomic, transcriptomic and epigenetic analyses linked GLUT3-dependent glucose utilization to mitochondrial metabolism and histone acetylation. We identified ATP-citrate lyase (ACLY), which catalyzes the release of cytosolic acetyl-CoA from mitochondrial citrate, as a druggable metabolic checkpoint downstream of GLUT3 to mitigate the pathogenicity of inflammatory T cells. Our work revealed a novel GLUT3-dependent metabolic-epigenetic circuit that controls the effector function of T cells but several important questions remain:
- What are the molecular and functional differences between the two glucose transporters in leukocytes?
- How is glucose metabolism is spatially organized, e.g., by directed metabolic channeling or subcellular compartmentalization?
- Is glucose transport and metabolism regulated in a cell and tissue-dependent manner?
- Can we exploit nutrient transporters to engineer metabolically optimized (CAR) T cells to treat malignancies and other immune-related pathologies?
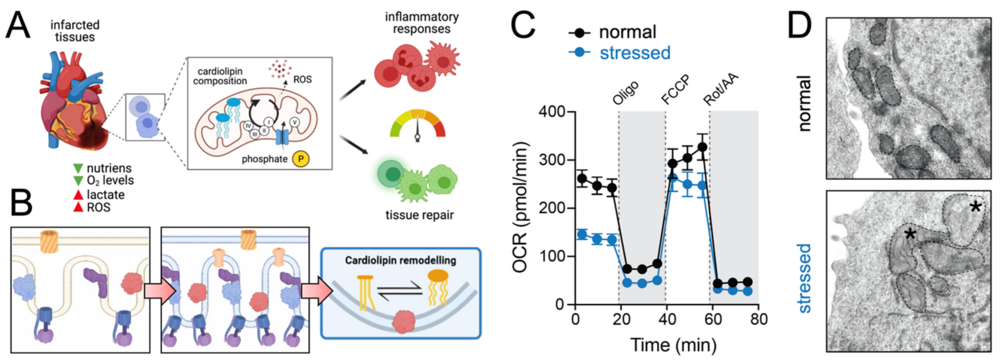
Mitochondria play a pivotal role in cellular metabolism by producing chemical energy in the form of ATP. The respiratory chain complexes generate a proton gradient following oxidative phosphorylation (OXPHOS) and fatty acid oxidation (FAO) at the inner mitochondrial membrane that is utilized by the ATP synthase (complex V) to generate ATP from ADP and inorganic phosphate. In addition to the ‘textbook function‘ of the mitochondria, intermediates of the tricarboxylic acid (TCA) cycle control the fate and function of immune cells by regulating signaling pathways, cellular redox balance, apoptosis and metabolic-epigenetic rewiring through posttranslational DNA and histone modifications. Thus, mitochondrial metabolism plays an eminent role in the life (and death) of leukocytes. Our lab is specifically interested to untangle the importance of mitochondria as the cellular powerhouses (i.e. the production of ATP) and their non-energetic functions, such as regulating signal transduction and their role in epigenetic reprogramming. To address these questions, we generated a series of immune cell-specific research models to investigate the following mitochondrial functions:
- Mitochondrial phosphate handling a s a prerequisite for ATP production
- Cardiolipins and the composition of the inner mitochondrial membrane
- The role of mitochondria as subcellular calcium buffers
- Mitochondrial ROS and its importance as a secondary messenger in signal transduction
- Mitochondrial insufficiency as an accelerator of immunosenescence and aging
Using preclinical models of autoimmunity, viral infection, and myocardial infarction, we also explore the potential of mitochondrial metabolism as a novel target for the treatment of inflammatory diseases.
PUBLICATIONS
Complete publication list on Pubmed: https://www.ncbi.nlm.nih.gov/myncbi/1pyNMxsZw4B58/bibliography/public/
Selected Articles
Mitochondrial dysfunction promotes the transition of precursor to terminally exhausted T cells through HIF-1α-mediated glycolytic reprogramming. Wu, H., Zhao, X., Hochrein SM, Eckstein, M., Gubert GF, Knöpper, K., Mansilla, AM, Doucet-Ladevèze, R., Schmitz, W., Ghesquière, Theurich, S., Dudek, J., Gasteiger, G., Zernecke, A., Kobold, S., Kastenmüller, W., and Vaeth, M. (2023). Nat Commun 14, 6858. 10.1038/s41467-023-42634-3
The glucose transporter GLUT3 controls T helper 17 cell responses through glycolytic-epigenetic reprogramming. Hochrein, SM, Wu, H., Eckstein, M., Arrigoni, L., Herman, JS, Schumacher, F., Gerecke, C., Rosenfeldt, M., Grün, D., Kleuser, B., Gasteiger, G., Kastenmüller, W., Ghesquiere, B., Van den Bossche, J., Abel, ED and Vaeth, M. (2022). Cell Metab 34, 516-532 e511. 10.1016/j.cmet.2022.02.015.
Genetic ablation of the mitochondrial calcium uniporter (MCU) does not impair T cell-mediated immunity in vivo. Wu, H., Brand, B., Eckstein, M., Hochrein, SM, Shumanska, M., Dudek, J., Nickel, A., Maack, C., Bogeski, I., and Vaeth, M. (2021). Front Pharmacol 12, 734078. 10.3389/ fphar.2021.734078.
BATF3 programs CD8+ T cell memory. Ataide, MA, Komander, K., Knopper, K., Peters, AE, Wu, H., Eickhoff, S., Gogishvili, T., Weber, J., Grafen, A., Kallies, A., Garbi, N., Einsele, H., Hudecek, M., Gasteiger, G., Holzel, M., Vaeth, M. and Kastenmüller, W. (2020). Nat Immunol 21, 1397-1407. 10.1038/s41590-020-0786-2.
Tissue resident and follicular Treg cell differentiation is regulated by CRAC channels. Vaeth, M., Wang, YH, Eckstein, M., Yang, J., Silverman, GJ, Lacruz, RS, Kannan, K., and Feske, S. (2019). Nat Commun 10, 1183. 10.1038/s41467-019-08959-8.
Store-operated calcium entry controls clonal expansion of T cells through metabolic reprogramming. Vaeth, M., Maus, M., Klein-Hessling, S., Freinkman, E., Yang, J., Eckstein, M., Cameron, S., Turvey, SE, Serfling, E., Berberich-Siebelt, F., Possemato R. and Feske, S. (2017a). Immunity 47, 664-679 e666. 10.1016/j. immuni.2017.09.003.
ORAI2 modulates store-operated calcium entry and T cell-mediated immunity. Vaeth, M., Yang, J., Yamashita, M., Zee, I., Eckstein, M., Knosp, C., Kaufmann, U., Karoly Jani, P., Lacruz, RS, Flockerzi, V., Kacskovics I., Prakriya M. and Feske, S. (2017b). ORAI2 modulates store-operated calcium entry and T cell-mediated immunity. Nat Commun 8, 14714. 10.1038/ncomms14714.
Store-operated calcium entry in follicular T cells controls humoral immune responses and autoimmunity. Vaeth, M., Eckstein, M., Shaw, PJ, Kozhaya, L., Yang, J., Berberich-Siebelt, F., Clancy, R., Unutmaz, D., and Feske, S. (2016). Immunity 44, 1350-1364. 10.1016/j. immuni.2016.04.013.
Selective NFAT targeting in T cells ameliorates GvHD while maintaining antitumor activity. Vaeth M., Bäuerlein CA, Pusch T., Findeis J., Chopra M., Mottok A., Rosenwald A., Beilhack A. and Berberich-Siebelt F. Proc Natl Acad Sci USA 112(4):1125-30. 10.1073/pnas.1409290112
Follicular regulatory T cells control humoral autoimmunity via NFAT2-regulated CXCR5 expression. Vaeth, M., Muller, G., Stauss, D., Dietz, L., Klein-Hessling, S., Serfling, E., Lipp, M., Berberich, I., and Berberich-Siebelt, F. (2014). J Exp Med 211, 545-561. 10.1084/jem.20130604.
Selected Review Articles
CRAC channels and calcium signaling in T cell-mediated immunity. Vaeth M, Kahlfuss S, Feske S. (2020). Trends Immunol (10):878-901. 10.1016/j.it.2020.06.012.
Calcium regulation of T cell metabolism. Wang Y, Tao A, Vaeth M, Feske S. (2020). Curr Opin Physiol 17:207-223. 10.1016/j.cophys.2020.07.016
NFAT control of immune function: New Frontiers for an Abiding Trooper. Vaeth M, Feske S. (2018) F1000 Res 2;7:260. 10.12688/f1000research.13426.1.
Ion channelopathies of the immune system. Vaeth M, Feske S. (2018) Curr Opin Immunol 52:39-50. 10.1016/j.coi.2018.03.021.
DFG project, VA 882/3-2 “Induction of NFATc1 controls the fate of activated T cells”
SFB 1526, project B06 “PANTAU – Targeting mitochondrial metabolism in pemphigoid diseases”
DFG project, VA 882/2-1 “The brain-type glucose transporter GLUT3 controls the function of regulatory T cells”