An engineered membrane-bound guanylyl cyclase with light-switchable activity
19.03.2021An engineered membrane-bound guanylyl cyclase with light-switchable activity (2021)
Yuehui Tian, Georg Nagel and Shiqiang Gao
BMC Biology volume 19, 54
Backround
Microbial rhodopsins vary in their chemical properties, from light sensitive ion transport to different enzymatic activities. Recently, a novel family of two-component Cyclase (rhod)opsins (2c-Cyclop) from the green algae Chlamydomonas reinhardtii and Volvox carteri was characterized, revealing a light-inhibited guanylyl cyclase (GC) activity. More genes similar to 2c-Cyclop exist in algal genomes, but their molecular and physiological functions remained uncharacterized.
Results
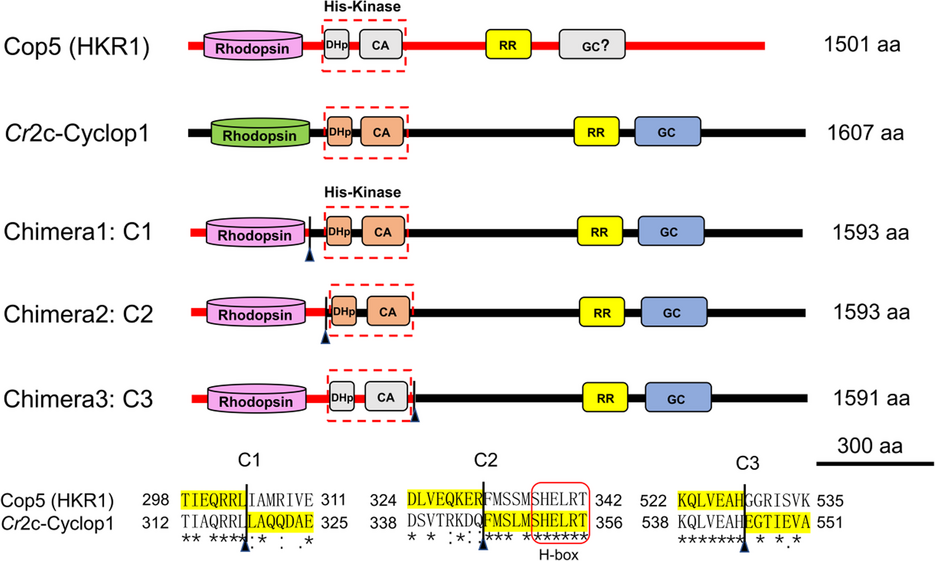
Chlamyopsin-5 (Cop5) from C. reinhardtii is related to Cr2c-Cyclop1 (Cop6) and can be expressed in Xenopus laevis oocytes, but shows no GC activity. Here, we exchanged parts of Cop5 with the corresponding ones of Cr2c-Cyclop1. When exchanging the opsin part of Cr2c-Cyclop1 with that of Cop5, we obtained a bi-stable guanylyl cyclase (switch-Cyclop1) whose activity can be switched by short light flashes. The GC activity of switch-Cyclop1 is increased for hours by a short 380 nm illumination and switched off (20-fold decreased) by blue or green light. switch-Cyclop1 is very light-sensitive and can half-maximally be activated by ~ 150 photons/nm2 of 380 nm (~ 73 J/m2) or inhibited by ~ 40 photons/nm2 of 473 nm (~ 18 J/m2).
Conclusions
This engineered guanylyl cyclase is the first light-switchable enzyme for cGMP level regulation. Light-regulated cGMP production with high light-sensitivity is a promising technique for the non-invasive investigation of the effects of cGMP signaling in many different tissues.